Submitted by Caralynn Wilczewski from Dr. Frank L Conlon's lab out of University of North Carolina, Chapel Hill.
Quantifying embryonic cardiac function in mammalian model systems of congenital heart disease (CHD) remains an outstanding challenge in the field of cardiovascular development. While these genetic models of CHD present compelling evidence of altered morphology, the functional consequences on cardiac function remain relatively unknown. Nevertheless, internally developing mammalian models remain popular research subjects for their homology to human cardiac development. While some ex vivo or invasive techniques have been developed to capture alterations in cardiac function, these techniques manipulate the endogenous developmental context and do not faithfully capture embryonic cardiac function in situ.
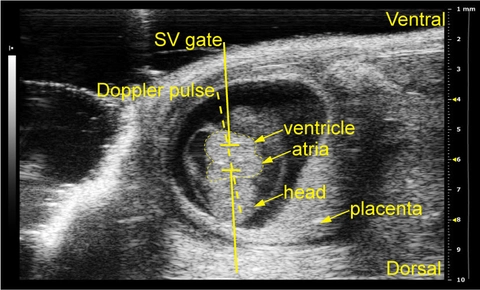
Using the Vevo 2100 and ultrasound pulsed-wave (PW) Doppler imaging, I have developed a reliable method for performing non-invasive in utero embryonic echocardiography on early gestation mouse embryos at embryonic day (E)10.5. In a model of non-cardiac myofibril gene misexpression, preliminary data indicates that mutant embryos show decreased cardiac function as measured by peak velocity and VTI compared to littermate controls. These results represent a shift in the methods by which embryonic models of cardiac development are assessed, and demonstrate a future for further reliance on in vivo measurements of altered cardiac function in models of cardiovascular development.
Image (top) shows schematic figure showing the approach to the Dopplers in an embryonic day (E)10.5 embryo in the dam’s abdomen that highlights the location of the heart.